 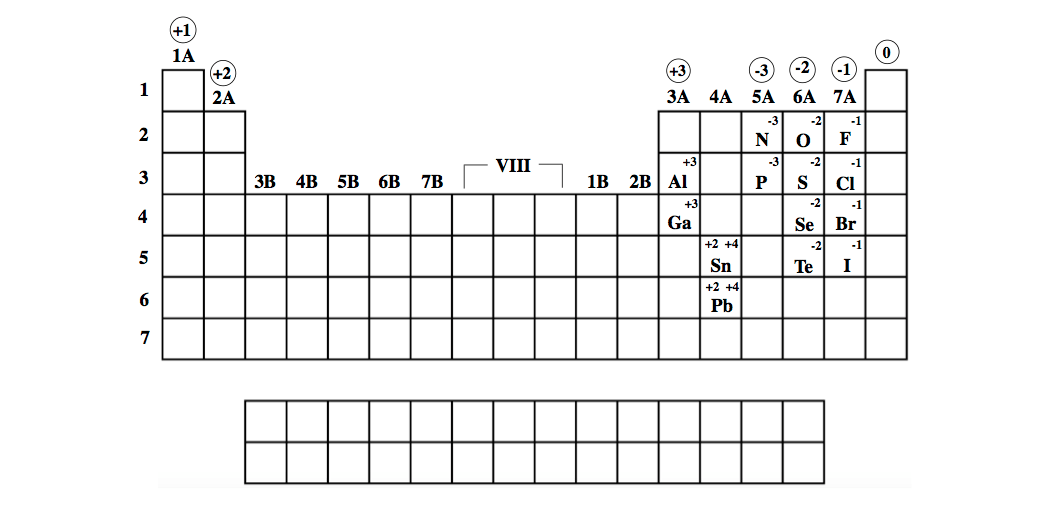
Since electrons are repelled by the negative charge, the cathode is seen as. When atoms (typically metals) lose one or more electrons the become positively charged cations. Ion-selective electrodes (ISEs) are electrochemical ion sensors that convert. These different types of ion forming metals will be discussed more in the next section on naming compounds ( section 2.7.3). Third, there are two types of metals, those that have only one charge state (like the nonmetals), and those that have multiple charge states. 
Give the names of the anion in each of the following compounds: CuS, Ag2SO4, Al (ClO3)3, Co (OH)2, PbCO3. To write the configuration for the Silver and the Silver ion, first we need to write the electron configuration for just Silver (Ag). Give the charges of the cation in each of the following compounds: CuS, Ag2SO4, Al (ClO3)3, Co (OH)2, PbCO3. Second, the nonmetals have only one charge state, which represents the number of electrons that need to be added for them to have the same number as a noble gas. Give the names of the cation in each of the following compounds: CuS, Ag2SO4, Al (ClO3)3, Co (OH)2, PbCO3. First, metals tend to from cations and nonmetals tend to form anions, while the noble gasses do not tend to form ions. There are several things you can note from this table though. In some systems with high ion activity (e.g., saline soils, fertilizer bands), net adsorption may be limited by the capacity of the PRS® Probe to hold ions.\) should not be considered to be a comprehensive Figure and this topic will be brought up again in the next section. Dichoroargentate(1-) AgCl2- CID 177710 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities. Ions of Some Main-Group Metals (Groups IA. The cation is given the same name as the neutral metal atom. Conversely, declining activities of a strongly held ion may not result in the amount adsorbed by PRS® Probes unless the activity and affinity of other ions is sufficient to displace the strongly held ion. We have 3 - on this side we've got to have three plus on this side so what's the charge on iron well there is one iron ion in this compound. The charge on the cation is the same as the group number. Increasing activity of an ion that is strongly held will generally increase the quantity of that ion on PRS® Probes and reduce the quantity of ions that are weakly held. The principles of ion exchange continue to apply. Soils are constantly undergoing change, and therefore the quantity of ions adsorbed on PRS® Probes may also change. So, we can write that the charge on chromium ions will be- 2+2x-140 So, solving this we will get that charge on each chromium ion. So, in total it will have a charge of -14. We have 7 oxygen atoms and each has a charge of -2. Here, we can see that we will have 2 silver ions. We will assume that all of the metallic elements other than those mentioned above can have more than one charge, so their cation. Within a given valence, ions that have a smaller dehydrated radius are generally held more strongly. In the question, it is given to us that charge on silver ions is +1. Relative affinity of an ion on a resin membrane (or other soil colloids) is most strongly influenced by valence: divalent ions are retained more strongly than monovalent ions, and trivalent ions are retained even more strongly. Cations are positively charged ions, and anions are negatively charged ions. 107 Ag est l’isotope le plus courant, avec une abondance naturelle d’environ 51. In both systems, a critical rate-limiting step is diffusion to the sink in response to gradients established by dynamic equilibria.Įventually, the quantity of a specific ion adsorbed on the ion exchange membrane often reaches a constant level that is Equilibrium Dependent, it depends on the relative activity in soil solution and relative affinity on the membrane of the ion. L’argent est présent dans 2 isotopes naturels: 107 Ag et 109 Ag. The process is similar to that occurring in the soil-root system, in which roots adsorb nutrients from soil solution by releasing counter ions such as H +, OH -, and HCO 3. When PRS® Probes are first inserted in soil, the rate of exchange is Diffusion Dependent: the flux of ions to the ion exchange membrane is controlled by the activity and diffusivity of ions in soil solution. 
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
